

How
a Slime Mold
Came to the Aid of
Alzheimer's Research
by Kathleen Cason
Intro/Proteins
run amuck
| The path to hirano bodies
Serendipity and beyond
|
What's ahead

(Top) Ten to 20 years before any indication of Alzheimer�s disease
is evident, the brain�s memory centers begin to shrink.
(Middle) As the damage spreads, an otherwise healthy person experiences
impaired memory, language and thinking.
(Bottom) During the final stages of Alzheimer�s disease, brain
atrophy is widespread and patients no longer recognize loved ones nor
have the ability to communicate.
For more information about Alzheimer�s disease, log on to www.alzheimers.org
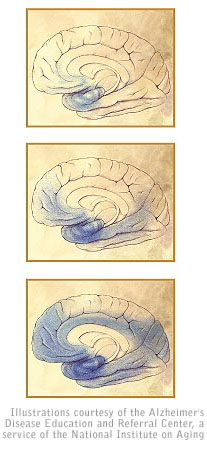
Blue shading indicates affected regions of the brain at various stages of
Alzheimer�s disease: preclinical (top), mild or moderate (middle) and severe
symptoms (bottom).
Intro
In an ancient Persian fable, three princes from the kingdom of Serendip go out into the world to gain experience. During their adventures, they find they have a gift for making unexpected discoveries.
Like those fabled princes, UGA cell biologist Marcus Fechheimer and his research team have discovered they too have a gift for serendipity.
In the course of trying to understand how proteins “know” where to go in a cell, the researchers unexpectedly stumbled upon an unusual cellular structure normally associated with Alzheimer’s and other neurodegenerative diseases.
“We didn’t expect to find a structure in a slime mold that
is present in increased amounts in people with Alzheimer’s —
particularly present in the brain’s major site of learning and memory.
That’s where these things accumulate,” he said.
Fechheimer’s team not only chanced upon this unusual structure —
called a Hirano body — in a slime mold but also figured out how to
force cells to produce them on command in the laboratory. Up till now,
these structures could only be studied in brain tissue of people who had
died. Because of this discovery, scientists will be able to study Hirano
bodies in living cells for the first time and unravel the mysteries of
this little-understood protein deposit.
Though Hirano bodies are common in the brains of people diagnosed with
dementia, their role in disease, if any, is unclear.
“We don’t know if a Hirano body is good or bad or why it’s
there or how it forms,” said Ruth Furukawa, a UGA research scientist
who co-directs the study.
Proteins run amuck What causes neurodegenerative diseases like Alzheimer’s is still
largely unknown, but something destroys nerve cells in the brain over
a period of time as victims gradually lose their minds.
In the hunt for cause and cure, scientists have focused on various abnormal
protein deposits that mar diseased brains. Deposits called plaques and
tangles have captured the most attention. Hirano bodies, while not at
the forefront of Alzheimer’s research, are another type of protein
deposit associated with the disease.
“Hirano bodies are certainly more prevalent in brains from patients
who are suffering from dementias, probably any type,” said James
Bamburg, one of Fechheimer’s collaborators and a professor of biochemistry
and molecular biology at Colorado State University. “Hirano bodies
also occur in brains of individuals with normal cognitive function but
usually increase in number with age.”
Nearly four decades have passed since Asao Hirano, an eminent neuropathologist
at Montefiore Medical Center in New York, first discovered the peculiar
deposit in the brain’s memory center. Since then Hirano bodies have
been reported in the brains of people with neurodegenerative diseases,
as well as diabetes, stroke and alcoholism.
“Making a Hirano body may be a cellular mechanism for dealing with
run-amuck proteins,” said Furukawa, who manages the day-to-day operation
of the lab. “That’s just a hypothesis.”
Run-amuck proteins certainly seem to play some role in all the diseases
where Hirano bodies are found.
“Perhaps Hirano bodies do nothing; perhaps they’re part of
cell death; or perhaps they are adaptations to stress that are good for
cells,” Fechheimer said. “Because they’re seen in so many
diseases, it’s worth finding out.”
After more than 20 years probing basic questions in cell biology, Fechheimer’s
team is well positioned to explore the role of Hirano bodies in cells
and in human disease.
“I didn’t think we were working on anything that had to do
with Alzheimer’s disease,” said Fechheimer, who uses a slime
mold as his lab’s version of a “guinea pig.”
Intro/Proteins
run amuck
| The path to hirano bodies
Serendipity and beyond | What's
ahead
For comments or for information please e-mail the editor: jbp@ovpr.uga.edu
To contact the webmaster please email: ovprweb@uga.edu
![]()